Boosting sterile packaging reliability for implantable medical devices
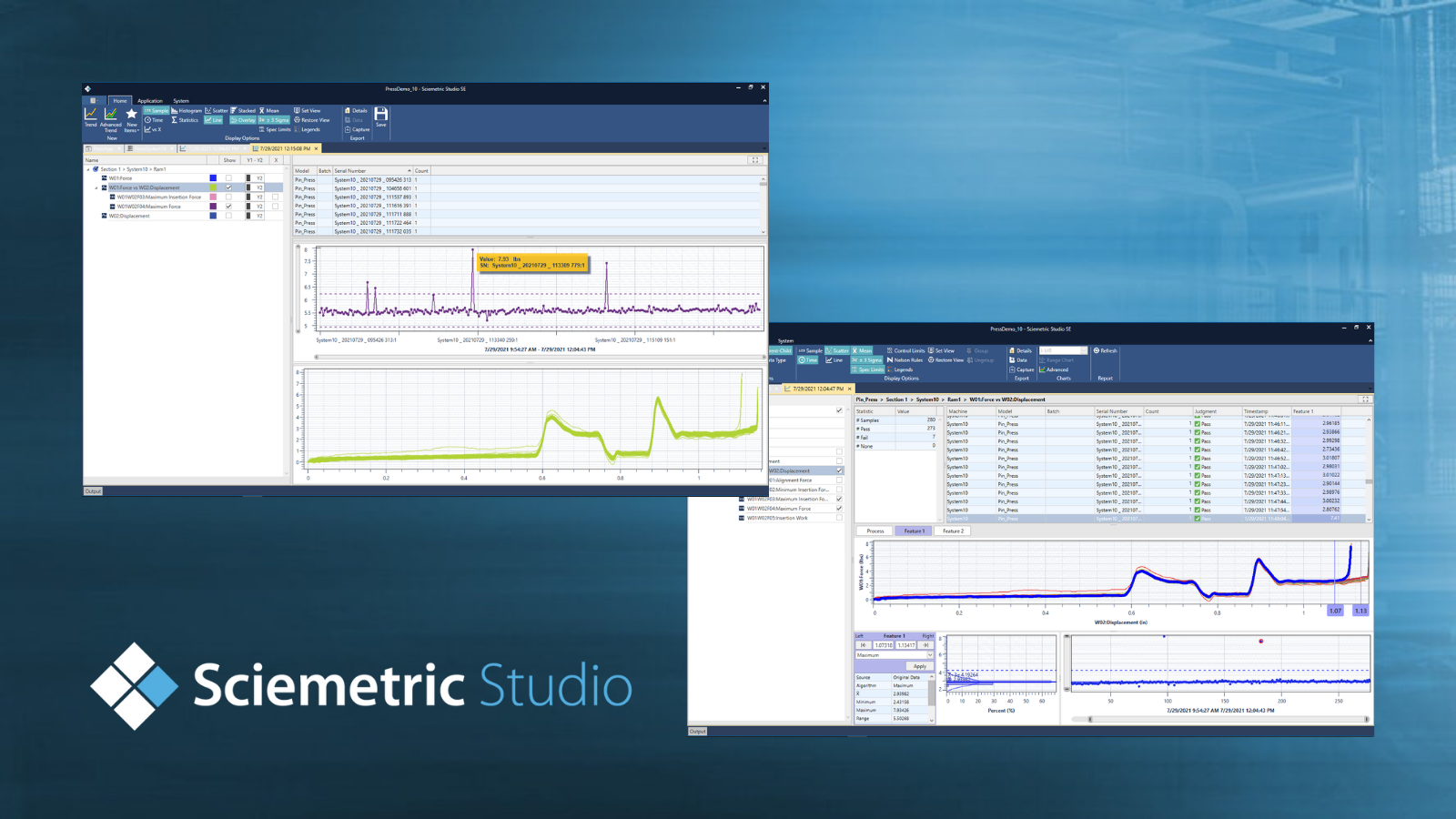
3 new ways to use Sciemetric Studio to improve your production line
Using data to contain the damage of a recall
The “R” word, recall, makes manufacturers cringe. Richard Brine digs into how the scope and impact of a recall can be dramatically reduced if all relevant production data has been collected and organized by serial number to create a complete birth history record for each part.
6 day-to-day benefits manufacturers can achieve with organized and correlated part production data
Using NVH monitoring to identify faulty axle snap ring installations—catch the problems manual checks miss
How to cut production downtime using data you’re already collecting
Integrators Part 2: Adapt to a changing market and avoid the ‘Nedry Effect’
Remember that Jurassic Park character, Nedry, who built an IT system no one else could manage? Joe Ventimiglio discusses how machine builders can avoid this mess by not trying to scratch-build a data-driven quality assurance system for a customer.
Different Types of Manufacturing Data Systems (Overview)
5 ways to make a manufacturing engineer’s life easier
Seeing the unseeable – winning the war against bubbles
Even the tiniest air bubbles can ruin the integrity of a seal or adhesive and vision systems alone can’t spot trapped air. Aaron Alberts talks about how Sciemetric teamed with Graco to create a new data-driven quality assurance system for dispense to capture what a camera can’t.